02.04.2026
Forschende aus Cambridge und Dresden zeigen einen neuen molekularen Mechanismus des Tumorwachstums
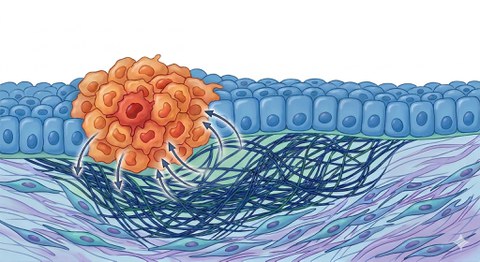
Frühe Tumorzellen verändern das umliegende Stützgewebe so, dass eine Umgebung entsteht, die ihr Überleben und weiteres Wachstum erleichtert. In diesen Prozess sind Bindegewebe, Fibroblasten und Immunzellen aktiv eingebunden.
Mit zunehmendem Alter sammeln sich im gesunden Gewebe Mutationen, die Krebs auslösen können. Allerdings entwickeln sich nur wenige dieser veränderten Zellen zu Tumoren. Erkenntnisse moderner Studien deuten darauf hin, dass die Entstehung von Tumoren von komplexen Wechselwirkungen zwischen den mutierten Zellen und ihrer Umgebung abhängt. Das Verständnis dieser Prozesse und der dazugehörigen Umweltfaktoren ist bislang jedoch begrenzt. Ein internationales Forschungsteam unter der Leitung der Universität Cambridge und mit Beteiligung der Medizinischen Fakultät der TUD sowie des Max-Planck-Instituts für molekulare Zellbiologie und Genetik (MPI‑CBG) hat nun einen bislang unbekannten Mechanismus beschrieben, der das Überleben von Tumorzellen in einem sehr frühen Stadium fördert.
Die kürzlich in Nature veröffentlichte Studie untersucht, warum manche mikroskopisch kleinen Tumoren im Gewebe unmittelbar nach ihrer Entstehung wieder verschwinden, während andere bestehen bleiben und später zu Krebs fortschreiten. Um diese Frage zu beantworten, modellierte das Team unter der Leitung von Dr. Maria P. Alcolea (Cambridge Stem Cell Institute) die frühen Stadien von Krebserkrankungen in der Speiseröhre von Mäusen. Die Forschenden konnten dabei die wenigen Tumoren identifizieren, die den natürlichen Schutzbarrieren des Gewebes entkommen, und analysieren, was sie von den vielen anderen unterscheidet, die frühzeitig eliminiert werden. Der Fokus lag dabei auf dem Epithelgewebe, das äußere und innere Körperoberflächen bedeckt und schützt.
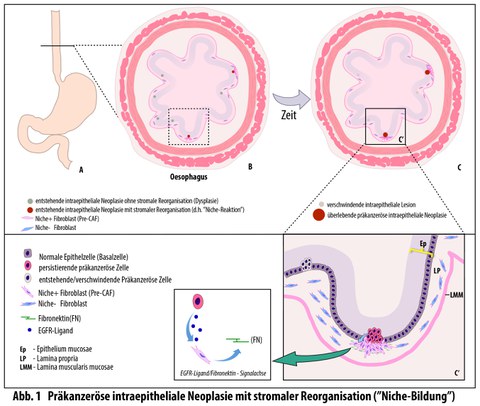
Die meisten neu entstehenden Tumoren in der Speiseröhre werden kurz nach ihrer Bildung durch Konkurrenz aus dem Gewebe verdrängt. Die aktuelle Studie zeigt jedoch, dass neue epitheliale Tumorzellen unmittelbar nach ihrer Entstehung Stresssignale an das darunterliegende Gewebe senden. Diese Signale aktivieren – ähnlich wie bei Wundheilungsprozessen – Fibroblasten der Lamina propria, einer dünnen Bindegewebsschicht. Die aktivierten Fibroblasten bilden ein Schutzgerüst um die anfänglich intraepitheliale Läsion, die noch auf die oberste Zellschicht begrenzt ist. Auf diese Weise entsteht eine besondere Mikroumgebung, die als „präkanzeröse Nische“ bezeichnet wird und das Überleben und Wachstum des Tumors fördert.
Auf molekularer Ebene beruht dieser Mechanismus auf einer EGF‑SOX9‑FN1‑Signalachse, die Wachstum und Verhalten der Zellen steuert. Unterbanden die Forschenden die molekulare Kommunikation zwischen den beiden Gewebestrukturen, bildete sich die präkanzeröse Nische nicht aus, und deutlich weniger frühe Tumoren überlebten.
Untersuchungen von menschlichem Gewebe bestätigten die Ergebnisse: Auch hier fanden sich frühe Tumorzellen mit charakteristischen Stresssignalen und dem gleichen fibrotischen Gerüst. Diese Erkenntnisse könnten die Früherkennung von Speiseröhrenkrebs verbessern, denn diese Krebsart wird oft erst in einem späten Stadium entdeckt, wenn die Behandlungsmöglichkeiten bereits eingeschränkt sind.
An der Studie waren Prof. József Jászai und Prof. Mirko HH Schmidt vom Institut für Anatomie der Medizinischen Fakultät Carl Gustav Carus der TUD sowie Prof. Daniela Aust vom Institut für Pathologie des Universitätsklinikums Dresden beteiligt. Die Dresdner Arbeitsgruppen stellten menschliches Gewebematerial von Patient:innen mit Speiseröhrenkarzinomen zur Verfügung und führten die dazugehörigen pathologischen Untersuchungen durch. Die Proben wurden sorgfältig ausgewählt, aufgearbeitet und histologisch sowie immunhistologisch prozessiert, um Tumor- und gesundes Gewebe zu vergleichen. Damit trugen die Dresdner Teams dazu bei, die im Mausmodell entdeckten Mechanismen im menschlichen Gewebe zu bestätigen und ihre Bedeutung für frühe Stadien des Speiseröhrenkrebses zu belegen.
Die Forschungsergebnisse zeigen, dass nicht nur genetische Veränderungen darüber entscheiden, ob ein Tumor überlebt, sondern auch das Zusammenspiel zwischen den veränderten Zellen und ihrer unmittelbaren Umgebung. Die charakteristischen „präkanzerösen Nischen“ könnten künftig als frühe biologische Hinweise dienen, um entstehende Tumoren schneller zu erkennen.
Prof. Esther Troost, Dekanin der Medizinischen Fakultät, sagt: „Die Identifizierung präkanzeröser Nischen als Voraussetzung für das Überleben früher Tumorzellen ist ein wichtiger Befund für künftige Krebstherapien. Dass Dresdner Forschungsteams diese Mechanismen im menschlichen Gewebe bestätigen konnten, stärkt unseren onkologischen Schwerpunkt und zeigt die besondere Expertise unseres Standorts in der translationalen Krebsforschung.“
Originalpublikation:
Skrupskelyte, G. et al. Precancerous niche remodelling dictates nascent tumour persistence. Nature; 4 March 2026; DOI: 10.1038/s41586-026-10157-8.
Hintergrund
Die Medizinische Fakultät Carl Gustav Carus der Technischen Universität Dresden (TUD) gehört zu den jüngsten und dynamischsten Fakultäten Deutschlands und setzt mit Schwerpunkten in Onkologie, Stoffwechsel- sowie neurologischen und psychischen Erkrankungen nationale und internationale Maßstäbe. Gemeinsam mit dem Universitätsklinikum Carl Gustav Carus bildet sie die Hochschulmedizin Dresden, einen führenden Standort für patientenorientierte Forschung und moderne Medizin.
Das Institut für Anatomie an der Medizinischen Fakultät erforscht molekulare Signaltransduktionsprozesse, die durch Proteine vermittelt werden, welche von Zellen freigesetzt werden oder in deren Plasmamembran lokalisiert sind. Das Institut arbeitet mit einem breiten Spektrum biowissenschaftlicher Methoden, von biochemischen Analysen über moderne Bildgebung und In-vivo-Modelle bis hin zur Analyse menschlicher Proben. Das Institut für Pathologie am Universitätsklinikum Dresden untersucht jährlich rund 48.000 Gewebeproben aus operativen und nichtoperativen Bereichen. Mit histologischen, immunhistologischen und molekularpathologischen Verfahren deckt das Institut das gesamte Spektrum der bioptischen und autoptischen Diagnostik ab und ist ein zentraler Partner der klinischen Fächer.
Wissenschaftlicher Kontakt:
Prof. József Jászai
Institut für Anatomie der Medizinischen Fakultät Carl Gustav Carus
an der Technischen Universität Dresden
Tel.: +49 (0) 351 458 6085
E-mail:
Kontakt für die Medien:
Anne-Stephanie Vetter
Stabsstelle Öffentlichkeitsarbeit
Medizinische Fakultät Carl Gustav Carus
der Technischen Universität Dresden
Tel. +49 (0) 351 458 17903
E-Mail: