Collaboration Claudia Arndt & Anja Feldmann & Anke Fuchs
Off-the-Shelf Adapter CAR-T cells for Hematological Disorders

Claudia Arndt, Anke Fuchs & Anja Feldmann
Recent advancements in cell-based immunotherapies, particularly switchable adapter chimeric antigen receptor (CAR) T cells, hold great promise for personalized cancer therapies. CAR-T cells are typically engineered from the patient's own T cells. Although autologous therapy is successfully applied, it frequently encounters challenges that significantly limit its broad applicability in clinical practice. These include substantial economic expenses, diminished T cell fitness in cancer patients, and restricted accessibility due to factors such as long production times or insufficient T cell quantities. Thus, readily available universal "off-the-shelf" CAR-T products with enhanced safety profiles are urgently needed to facilitate broad applicability across all cancer patients with no waiting time and reduced costs.
Claudia Arndt and Anja Feldmann are experts in T cell and antibody engineering and made significant contributions to the preclinical development of the adaptor CAR-T technologies "UniCAR" and "RevCAR", which successfully entered first-in-human clinical trials (NCT04230265, NCT05949125). Building on over 10 years of expertise, they are aiming to combine CAR‑T cell therapy with targeted radiotherapy and in vivo imaging to design more powerful personalized cancer theranostics.
Anke Fuchs, research group leader (Advanced Cellular Therapeutics lab) at the Center for Regenerative Therapies Dresden (CRTD) and scientific lead of the CRTD GMP facility, is an immunologist with over 10 years of experience in clinical translation of Advanced Therapy Medicinal Products (ATMP). She developed automated manufacturing processes and holds manufacturing licences for regulatory T and effector T cell based products.
Anja Feldmann and Anke Fuchs are Principal Investigators and working group leaders within the SaxoCell Cluster4Future (https://www.saxocell.de).
Research aims
Our research mission is to manufacture a controllable, "off-the-shelf" CAR-T cell product derived from umbilical cord blood with an improved safety profile that is readily available at low cost to any cancer patient.
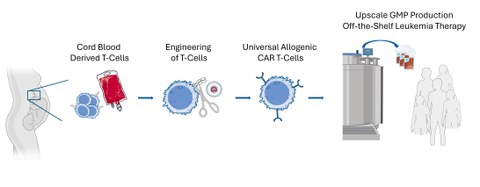
© Created with bioart.niaid.nih.gov/bioart/421 & biorender.com
Our specific research aims include:
- Development of a manufacturing protocol for edited allogeneic UniCAR-T cells derived from frozen umbilical cord blood (UCB)
- Implementation of a GMP-compliant upscaled process toward a manufacturing license
- State-of-the-art tracking of allogeneic UCB UniCAR T cells and tumor targets using multimodal imaging in preclinical models
Our vision:
- Preparation for a First-in-Human clinical trial evaluating allogeneic UCB UniCAR-T cell therapy for T-cell acute lymphoblastic leukemia (T-ALL)
Members

Dr. Claudia Arndt
Dr. Claudia Arndt
Research Group Leader MSNZ
Helmholtz-Zentrum Dresden-Rossendorf (HZDR)
phone: +49-351-458-4049

Dr. Anja Feldmann
Dr. Anja Feldmann
Head of Department MSNZ
Helmholtz-Zentrum Dresden-Rossendorf
phone: +49-351-458-3428

Dr. Anke Fuchs
Dr. Anke Fuchs
Research group Leader MSNZ
Center for Regenerative Therapies Dresden (CRTD)
phone: +49-351-458-82125

Dr. Samikshya Santosh Nirmala
Dr. Samikshya Santosh Nirmala
Postdoc MSNZ
Center for Regenerative Therapies Dresden (CRTD)
phone: +49-351-458-82110

Dr. Jurie Tashkandi
Dr. Jurie Tashkandi
PostDoc MSNZ
Helmholtz-Zentrum Dresden-Rossendorf
phone: +49-351-458-3428

Msc. Lamice Shurafa
Msc. Lamice Shurafa
PhD student MSNZ
Helmholtz-Zentrum Dresden-Rossendorf
Selected publications
Santosh Nirmala S, Hu Y, Floegel FD, Cruz H, Morgenstern J, Platz A, Vollroth M, Fuchs, A. (2026). Delineating phenotypic heterogeneity in human regulatory T cells across developmental stages and therapeutic sources. Frontiers in Immunology. (Vol. 17). https://doi.org/10.3389/fimmu.2026.1697723
Lakshmi K, Jutrzenka-Trzebiatowski A von, Loureiro L, Soto KEG, Peter K, Morales JMM, Nirmala SS, Berndt N, Arndt C, Hu Y, Li JW, Peitzsch C, Taubenberger A, Wehner R, Schmitz M., Hölig K, Abken H, Bonifacio E, Bornhäuser M, Bachmann M, Feldmann A, Fuchs A. (2025). Automated GMP-compatible production of universal CAR Tregs for organ-targeted tolerance induction. Journal of Translational Medicine, 23(1), 1399. https://doi.org/10.1186/s12967-025-07431-0
Peschke JC, Bergmann R, Mehnert M, Gonzalez Soto KE, Loureiro LR, Mitwasi N, Kegler A, Altmann H, Wobus M, Máthé D, Szigeti K, Feldmann A, Bornhäuser M, Bachmann M, Fasslrinner F, Arndt C. FLT3-directed UniCAR T-cell therapy of acute myeloid leukaemia. Br J Haematol. 2023 Sep;202(6):1137-1150. doi: 10.1111/bjh.18971. Epub 2023 Jul 17. Erratum in: Br J Haematol. 2024 Mar;204(3):1120. doi: 10.1111/bjh.19328. PMID: 37460273.
Arndt C, Bergmann R, Striese F, Merkel K, Máthé D, Loureiro LR, Mitwasi N, Kegler A, Fasslrinner F, González Soto KE, Neuber C, Berndt N, Kovács N, Szöllősi D, Hegedűs N, Tóth G, Emmermann JP, Harikumar KB, Kovacs T, Bachmann M, Feldmann A. Development and Functional Characterization of a Versatile Radio-/Immunotheranostic Tool for Prostate Cancer Management. Cancers (Basel). 2022 Apr 14;14(8):1996. doi: 10.3390/cancers14081996. PMID: 35454902; PMCID: PMC9027777.
Kittel-Boselli E, Soto KEG, Loureiro LR, Hoffmann A, Bergmann R, Arndt C, Koristka S, Mitwasi N, Kegler A, Bartsch T, Berndt N, Altmann H, Fasslrinner F, Bornhäuser M, Bachmann MP, Feldmann A. Targeting Acute Myeloid Leukemia Using the RevCAR Platform: A Programmable, Switchable and Combinatorial Strategy. Cancers (Basel). 2021 Sep 24;13(19):4785. doi: 10.3390/cancers13194785. PMID: 34638268; PMCID: PMC8508561.
