A7
Brain states representing dynamics of meta-control
In cognitive control (e.g. choice reaction time tasks with distractors) or decision making tasks (e.g. intertemporal choices, when subjects need to choose between an immediate reward and a larger delayed reward), the behavior of a subjects is often summarized by one parameter that is thought to reflect, for example, the emotional reactivity or the impulsivity of the subject. Psychologists refer to this as a subject or personality trait. However, such traits may not necessarily be stable over time but rather dependent on meta-control states that alter subject behavior in different ways.
It is the goal of this project to study the nature of such transient control states, especially in terms of brain activity and connectivity as measured by functional magnetic resonance imaging (fMRI), and to investigate factors and methods that may be used to intentionally induce such states. Such interventions may subsequently be used to treat pathological behavior or research the causes for a particular behavior.
Concretely, we examine:
- how emotional stimuli effect our cognitive abilities,
- whether neurofeedback can be used to influence emotional reactions,
- how brain stimulation changes connectivity of emotional networks,
- whether predictions of intertemporal choices can be improved when considering trial-specific brain activity,
- whether a current attentional shielding state can be detected on the basis of brain activity, and
- whether detection of particular brain states in possible in real-time to manipulate an ongoing task towards a desired goal (e.g. stronger attentional shielding).
Recent Findings:
Dynamically changing cognitive control states influence our ability to respond to our environment in a controlled manner. With respect to emotional control states and their related brain states, we showed, for example, that baseline activity of the amygdala, a brain region responsible for emotional relevance detection, predicts reaction times (Riedel et al., 2016). We also showed that negative distractor images slow down reactions compared to neutral images and could relate this to the differential response of the insula to the emotional distractors (see Fig. 1).
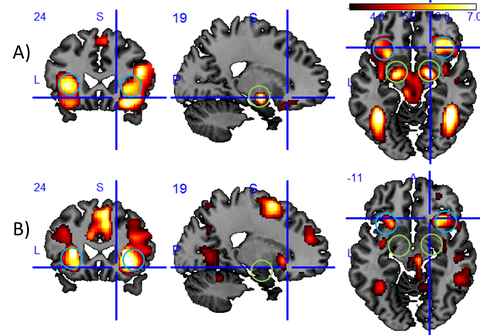
Figure 1: Results of an fMRI study of subjects that are asked to do a simple reaction time task while exposed to negative, neutral, or positive distractor images. In this task, reactions are slower for negative distractors than for neutral or positive distractors. Displayed is the brain activity (Group activation maps at MNI = (19, 24, -11) (N=40, P < 0.001 uncor.)) measured through the blood oxygen level-dependent (BOLD) effect for two different analysis on top of a conventional T1-contrast image: A) stronger activation for negative than for neutral distractor imagesl (T(117) > 3.16) and B) activation, which is positively correlated with reaction time (T(39) > 3.31). Blue circles mark the anterior insula, green circles the amygdala, two core brain regions involved in the processing of emotions. The fact that the insula shows up in both analyses suggests that this region may be a mediator of the stronger attentional capture effect by negative images as compared to neutral images. As the amygdala activation appears to be unrelated to the reaction time, we see no evidence in this data for the hypothesis that the attentional capture effect is driven by the amygdala.
We have also shown that interventional methods can alter brain states. Amygdala activity could be altered through fMRI neurofeedback training without an instructed regulation strategy (Marxen et al., 2016) and also through emotional imagery (Fig. 2). Furthermore, the functional connectivity of the frontal pole / superior frontal gyrus with the amygdala could be modulated differentially by inhibitory and excitatory transcranial magnetic stimulation (Riedel et al., in revision), a technique that is clinically used to treat depression. In this context, we also showed that amygdala function is altered in healthy adolescents with a family liability for depression (Pilhatsch et al., 2014).
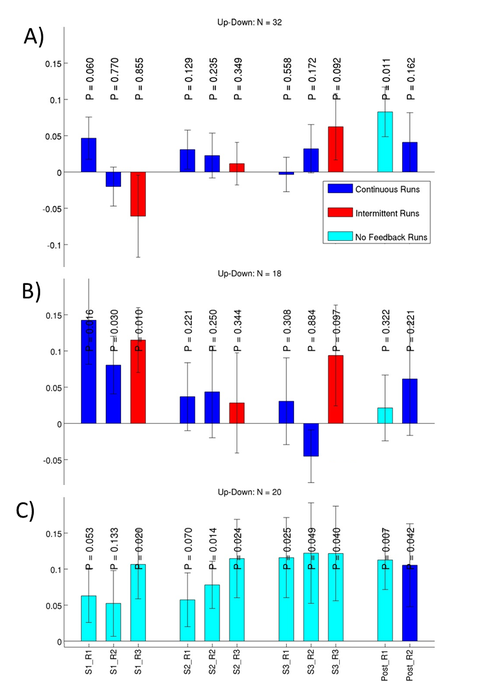
Figure 2: This plot illustrates the ability of subjects to willfully regulate brain activity in the amygdala, a brain region important for the perception of emotional stimuli. Each bar indicates the difference in brain activity in % when subjects are instructed to upregulate compared to down regulation. Three training ses-sions S1-S3 with 3 training runs R1-R3 each and a transfer run Post_R1 without neurofeedback plus a normal feedback run Post_R2 were conducted. P-values for one-sided t-tests are provided. Three differ-ent groups of subjects from two different studies are shown: (A) a group that received no suggested regulation strategy, (B) a group that was instructed to use “Positive Imagery” to up-regulate and “Mental Arithmatics” to down-regulate, and (C) a group that used the same regulation strategies as (B) but re-ceived no neurofeedback. The results of these studies show that subjects can regulate their brain activity after the training (Post_R1). However, there is no clear evidence that neurofeedback improves regulation abilities over and above employing a conventional regulation strategy. Because of small effect sizes, larger experimental groups are needed to examine the efficacy of neurofeedback.
Following the investigation of these emotional brain states, we investigated the possibility to detect brain states related to the Anticipation-Discounting Dilemma using multi-variate pattern analysis (MVPA) techniques. The dilemma was operationalized by an intertemporal choice (ITeCh) paradigm (Ripke et al., 2015). Using the same data, we could predict choices based on trial-by-trial value signals following offer presentation in the brain better than chance. However, the accuracies were much below prediction accuracies obtainable by conventional behavioral modeling (Knorr et al., draft). Additionally, adding the trial-by-trial brain data to the behavioral model would only degrade, not improve prediction accuracies. Furthermore, we conducted pilot experiments that showed that repeated presentation of ITeCh offers at the subject’s indifference point led to a subject-dependent change of the temporal discounting rate (unpublished results). Therefore, we concluded that our initial goal to detect trial-by-trial brain signals even before offer presentation, to predict subject choices at the indifference point based on these signals, and to associate such brain states with alterations in impulsivity as measured by behavior was not feasible.
Addressing these limitations, we began to investigate longer lasting, attentional brain states in the context of the Shielding-Shifting Dilemma. In a conventional random effects group analysis of set-switching data gathered by the B1 and B3 CRC Projects, we found an attention effect in expected regions in the contrast “target: faces; distractor: houses” versus “target: houses; distractor: faces”. To conduct an MVPA analysis, we modified the task, primarily extending attention block length, to test the hypothesis that shielding strength, as measured by the confidence of the MVPA classifier, will be negatively correlated with the behavioral interference costs. Twenty-five subjects have been acquired and are currently being analyzed. In these subjects, we will also investigate functional connectivity measures as classification features.
Project members
Principal Investigators
Michael Marxen
Group Leader Imaging Physics
Michael Smolka
Deputy Spokesperson CRC 940; Professor for Systems Neuroscience (W2)
Staff
Felix Knorr
Doctoral Researcher
Philipp T. Neukam
Postdoctoral Researcher
Publications
-
Marxen, M., Jacob, M. J., Muller, D. K., Posse, S., Ackley, E., Hellrung, L., . . . Smolka, M. N. (2016). Amygdala Regulation Following fMRI-Neurofeedback without Instructed Strategies. Frontiers in Human Neuroscience, 10. doi: 10.3389/fnhum.2016.00183
- Pilhatsch, M., Vetter, N., Hübner, T., Ripke, S., Müller, K.U., Marxen, M., Rodehacke, S., Mennigen, E., Schmidt, D., & Smolka, M.N. (2014). Amygdala-function perturbations in healthy mid-adolescents with familial liability for depression. Journal of the American Academy of Adolescent Psychiatry, 53(5), 559-568.e6. doi:10.1016/j.jaac.2014.02.010
-
Riedel, P., Jacob, M. J., Muller, D. K., Vetter, N. C., Smolka, M. N., & Marxen, M. (2016). Amygdala fMRI Signal as a Predictor of Reaction Time. Frontiers in Human Neuroscience, 10. doi: 10.3389/fnhum.2016.00516
-
Ripke, S., Hubner, T., Mennigen, E., Muller, K. U., Li, S. C., & Smolka, M. N. (2015). Common neural correlates of intertemporal choices and intelligence in adolescents. Journal of Cognitive Neuroscience, 27(2), 387-399. doi:10.1162/jocn_a_00698
