Feb 27, 2026
Harnessing the Power of AI to Uncover Hidden Changes in the Pancreases of People with Type 2 Diabetes
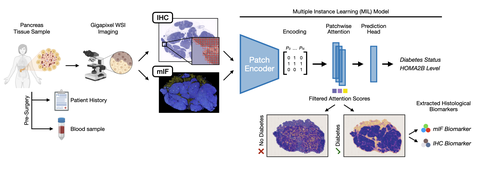
Workflow
Harnessing the Power of AI to Uncover Hidden Changes in the Pancreases of People with Type 2 Diabetes
A team of researchers from Germany and across the world have developed an explainable artificial intelligence (AI) pipeline to analyze high-resolution, whole-slide scans of pancreas tissue derived from human living donors to identify key morphological features linked to type 2 diabetes. By training deep learning models on a unique dataset of stained pancreas tissue, the explainable AI successfully detected subtle changes associated with type 2 diabetes, including islet -size, fat distribution, and fibrosis.
Type 2 diabetes is a major global health issue, affecting millions and placing a significant burden on healthcare systems. Failures in insulin—a key hormone that regulates blood glucose—function and production drive the disease. A key feature of this impairment is the failure of beta cells—the cells in the pancreas that secrete insulin. Despite extensive research, the exact cause and nature of the failure of beta cells remains unclear, but one clue might come from changes in morphological features of the pancreases in people with type 2 diabetes. However, because of the high degree of variability between individuals, it is very challenging for pathologists to distinguish between pancreatic tissue from a person with type 2 diabetes and those without.
Machine learning has attracted significant attention in the field of computational pathology due to its ability to analyze large volumes of information-rich, whole-slide images. Such algorithms can be as effective as expert pathologists. Up to now, most deep learning applications focus on oncological diseases, where distinct cancer cells are identifiable in tissue samples. In contrast, pancreatic tissue lacks clear quantifiable features that unequivocally differ between people with and without type 2 diabetes. The goal of the study was to identify if there are morphological features of pancreatic specimens that would enable a person with type 2 diabetes to be distinguished from one without diabetes and if so, reveal what those features are. Such a finding would help to understand the cause of diabetes better.
An interdisciplinary team including researchers from the German Cancer Research Center, (DKFZ), Institute for Diabetes Research and Metabolic Diseases (IDM), the Paul Langerhans Institute Dresden (PLID), and the German Diabetes Center (DDZ)/Heinrich Heine University Düsseldorf, as well as other institutes world-wide were brought together to achieve this goal. The team developed an explainable AI pipeline, which can, unlike conventional AI, show how it makes decisions. The researchers also collected a dataset of whole-slide images of pancreas tissue from living donors from the LIDOPACO cohort—the largest collection of pancreatic tissue from metabolically phenotyped living donors worldwide. These tissues were stained with various techniques to highlight different cell types and structures. A combination of deep learning and explainable AI models were trained on this dataset to distinguish between pancreatic tissue from persons with type 2 diabetes or without. The model was able to detect subtle differences in tissue morphology and thereby to predict type 2 diabetes with ~ 95% accuracy, thus outperforming pathologists. Surprisingly, the most effective predictions came from focusing on neuronal axons and the endocrine cells that produce glucagon and somatostatin—other hormones that regulate blood glucose—rather than the endocrine beta cells that produce insulin—the hormone that is typically associated with diabetes. This suggests that changes in these less-characterized cells may play a crucial role in type 2 diabetes pathology.
The study found several morphological features linked to type 2 diabetes. People with type 2 diabetes tend to have smaller islets, significantly larger fat cell clusters, and an increased amount of fibrosis. The explainable AI models also revealed that in tissue from people with type 2 diabetes, the distance between islets and fat cells was significantly shorter, suggesting that fat cells may be altering the pancreatic microenvironment.
The study’s findings underscore the complexity of type 2 diabetes, showing that the disease impacts not just the insulin-producing beta cells but also involves interactions between other cell types and subtle structural changes in the pancreas. This explainable AI-driven approach opens new doors for understanding type 2 diabetes, potentially leading to improved strategies for prevention, diagnosis, and treatment.
For more information visit the project website where the full dataset has been made available: https://diadem.dzd-ev.org
Original Article
Title: Explainable AI-based analysis of human pancreas sections identifies traits of type 2 diabetes. Journal: Nature Communications. 17, Article number: 1558 (2026). Athors: Klein L*, Ziegler S*, Gerst F*, Morgenroth Y*, Gotkowski K, Schöniger E, Heni M, Kipke N, Friedland D, Seiler A, Geibelt E, Yamazaki H, Häring HU, Wagner S, Nadalin S, Königsrainer A, Mihaljević AL, Hartmann D, Fend F, Aust D, Weitz J, Jumpertz-von Schwartzenberg R, Distler M, Maier-Hein K, Birkenfeld AL, Ullrich S, Jäger P**, Isensee F**, Solimena M**, Wagner R**. * These authors contributed equally. ** These authors jointly supervised this work. DOI: https://doi.org/10.1038/s41467-026-69295-2
