Latest news
International delegation visits BioBank Dresden
On April 14, 2026, we welcomed a high-ranking international delegation from Slovakia and the Czech Republic. The group of visitors consisted of representatives from a total of seven academic locations, including university hospitals, medical faculties and the national cancer institute.
The delegation was joined by industrial partners from the biobanking sector. The group had already made a stopover in Prague, after which the trip continued to Heidelberg, including a visit to NCT-Liquid and the DKFZ Repository.
During their stay in Dresden, our guests were given a comprehensive insight into the structures and working methods of BioBank Dresden. In addition to the respective biobank managers, numerous employees from various departments also took part, which enabled an intensive professional exchange. One of the aims of the visit was to gain inspiration for the further development and operation of professional biobanks in Slovakia and the Czech Republic. Although biobanks already exist in both countries, many processes are still highly manual and offer potential for further automation.
A special focus was placed on visiting the technical infrastructure of BioBank Dresden. During an extensive tour of the laboratory and storage areas, the existing automation solutions were presented in detail and their integration into daily workflows was explained. Our guests showed great interest in the technologies used, particularly in cell isolation systems, modern pipetting robots and automated cryotanks for the long-term storage of samples.
In addition to the pure presentation of the devices, the focus was on their practical application, the underlying process chains and aspects of standardization and quality assurance. This showed the significant contribution that automated systems can make to increasing efficiency, reducing errors and scalability in biobank operations. The solutions on display clearly illustrated how a high degree of process reliability, traceability and sample quality can be guaranteed through the use of modern technologies in a targeted manner.
In addition, there were lively discussions on a number of key topics relating to biobank operations. The focus here was on the range of samples, strategies for recruiting control samples, IT and database solutions, issues of broad consent and ethics, the linking of biosamples with clinical and analysis data and various financing models. Aspects such as personnel structures, accreditation and quality management were also discussed in depth.
Another important point of the exchange was the role of the German Biobank Network (GBN) and possible cooperation options at European level. The discussions underlined the great interest in closer international networking and cooperation. Further exchange, in particular with the GBN, is already being considered.
We would like to express our sincere thanks for the visit and the open, constructive dialog. Such international meetings make an important contribution to the further development of biobanks and the strengthening of joint research infrastructures in Europe.
-------------------------------------------------------------------------------------------------------------------------------------
BioBank Dresden strengthens research on long-COVID in children
On March 13, 2026, the Federal Minister of Health Nina Warken, the Saxon State Minister for Social Affairs, Health and Social Cohesion Petra Köpping and other political representatives visited the University Hospital Carl Gustav Carus Dresden to gain an insight into the PEDNET-LC project. The nationwide network unites 65 clinics and research institutions and aims to sustainably improve the diagnosis and treatment of children and adolescents with Long COVID and similar post-infectious diseases and to establish and ensure comprehensive care. The focus is on 20 specialized care centers that have been working together on an interdisciplinary basis since 2025 - including the Clinic for Pediatric and Adolescent Medicine in Dresden. The network is coordinated by Uta Behrends (Munich) and Nicole Töpfner (Dresden). The Federal Ministry of Health is funding PEDNET-LC over a period of four years with a total of 41 million euros.
We at BioBank Dresden are pleased to be part of the joint project and to be responsible for biobanking for the network as part of a work package together with the Freeze Biobank in Freiburg. Together we coordinate the provision of kits for sample collection, sample collection and shipping, processing, storage and long-term management of the pediatric biosamples. The biospecimens include a broad panel of blood, urine, hair, swab and/or stool samples. To date, almost 5200 biosamples from 264 patients have been submitted, and the goal for the funding period is to expand the inventory to 40,000 biosamples from 2000 patients. The aim is to continuously improve preventive, diagnostic and therapeutic strategies in the treatment of patients with PAIVS (Post-Acute Infection and Vaccination Syndrome) and ME/CFS (Myalgic Encephalomyelitis/ChronicFatigue Syndrome) and to deepen our understanding of the underlying pathophysiology. Our established infrastructure, which we were able to explain in a poster, convinced the ministers.
-------------------------------------------------------------------------------------------------------------------------------------
New animated film explains biobanking for children and families
The German Biobank Network (GBN), together with the University Hospital Carl Gustav Carus of TU Dresden and the DZKJ Leipzig/Dresden, has published an animated information film on the subject of biobanking for children and their guardians.
The video explains in an understandable and child-friendly way why biosamples are important for research, how they are used and how the protection of personal data is ensured. It supports families in making an informed and voluntary decision.
Even if there is no direct personal benefit, donors make a valuable contribution to medical research and future patients.
The film is now available for public relations work and to support educational talks.
-----------------------------------------------------------------------------------------------------------------------------
10 years NCT/UCC
The NCT/UCC Dresden looks back on ten years of cutting-edge research in which modern cancer medicine, innovative technologies and patient-oriented care have been closely interlinked. To mark the anniversary on December 15, Minister President Michael Kretschmer and numerous representatives from politics, science and the partner institutions involved visited the site and paid tribute to its outstanding role as a beacon of cancer medicine. The visit underlined the national importance of the center and the success of the Dresden model of transferring research results quickly and effectively into patient care.
BioBank Dresden was also represented with its own information stand and met with great interest from the many visitors to the anniversary event. The central role of biobanks for innovative, patient-oriented cancer research became clear in many intensive and stimulating discussions - including with the Minister President, representatives of patient advocacy groups, BMFTR employees, lawyers, researchers and doctors. The lively exchange impressively demonstrated the importance of close dialogue between research, clinics, politics and society in order to further strengthen the conditions for future progress in personalized medicine.
----------------------------------------------------------------------------------------------------------------------------------
Visit to Lifelines Biobank and LvL on October 13
On October 13, we had a visit from our partners Lifelines Biobank (NL) and LvL.
Hilde Laeremans and Rick Nijboer (Lifelines Biobank) as well as Waldemar Janzen and Nick Wolter (LvL) were with us.
Together, we took a closer look at the BBD, focusing in particular on our scanner technology and the integrated scanner solutions in the PBMC system. Exciting discussions focused on troubleshooting, robotic systems and the exchange of experiences and solutions. Our guests had a number of technical challenges to share their ideas and approaches with us.
Finally, there was a cozy evening event - a nice end to a day full of exciting impressions and discussions.
We would like to thank you for your visit and look forward to the next meeting!
----------------------------------------------------------------------------------------------------------------------------------
BioBank Dresden at the 13th National Biobank Symposium: Setting the course for a national biobank
From September 22 to 23, 2025, more than 200 experts met at the 13th National Biobank Symposium to discuss the future of biobanking in Germany. The focus was on the concept for a national biobank envisaged in the coalition agreement, which is to serve as a central digital platform to provide many millions of tissue and liquid samples as well as data for medical research.
As part of the Network of University Medicine (NUM), BioBank Dresden is supporting this pioneering project, which was jointly developed by a broad alliance of academic research and industry. The aim is to permanently secure access to quality-assured samples and to further improve the linking of biosamples with health data - especially in the course of digitalization and with a view to the European Health Data Space.
We see the National Biobank as a great opportunity to strengthen Germany as a research location and take biomedical research to a new level.
You can find more information here.
---------------------------------------------------------------------------------------------------------------------------------
Visit from Sweden: Teresa Mortera Blanco visits Biobank Dresden
We are delighted to have recently welcomed Dr. Teresa Mortera Blanco from the renowned Karolinska University in Stockholm to Biobank Dresden. Dr. Mortera Blanco heads the research department of the MDS working group of Prof. Eva Hellström-Lindberg - one of the leading international experts on myelodysplastic syndromes (MDS) and a member of the Nobel Prize Committee for Medicine. She is also responsible for the Karolinska Institute's hematology biobank, which has come into the international spotlight in recent months after a technical failure necessitated a comprehensive restructuring.
In an exciting lecture, Dr. Mortera Blanco presented the future concept of the new biobank, which includes measures to safeguard and avoid technical failures. In her search for innovative solutions, she came across our publication on automated PBMC processing using robotics - a focus of our work at Biobank Dresden. The technologies presented aroused great interest, and so the desire arose to get an idea of our infrastructure and processes on site.
Dr. Mortera Blanco was accompanied by representatives from Hamilton, who shared technical insights and details about our automation solutions during the tour. An inspiring exchange about common challenges, solutions and possible future forms of cooperation between the Dresden and Stockholm sites ensued.
We look back on an insightful day with valuable scientific dialogue and look forward to further exchanges with Dr. Mortera Blanco and the team at the Karolinska Institute.
---------------------------------------------------------------------------------------------------------------------------------
Our close cooperation partner GBN presents itself with a new name and logo.
All important information here
Kickoff of PEDNET-LC
Together with the Freeze Biobank we coordinate the Bioanking in Pednet-LC network. Biosamples are processed and stored according to common standards in either Freiburg or Dresden according to organized center assignments. They are linked to LC registry data and made available to Pednet-LC research.
It is a great pleasure and honor for us as BioBank Dresden to be part of this important project. The funding offers a unique opportunity to work together with partners from all over Germany to improve care and research on Long COVID and similar diseases in children and adolescents. We are proud to play our part in developing tailored, sustainable solutions for affected young people and advancing scientific collaboration in this important area.
---------------------------------------------------------------------------------------------------------------------------------
German Biobank Node becomes part of the University Medicine Network - we at BioBank Dresden are also involved
The German Biobank Node (GBN), which has been driving forward the networking and standardization of biobanks in Germany for years, will be integrated into the Network University Medicine (NUM) on 1 July 2025. This will further strengthen cooperation between biobanks and university research. In future, the GBN will operate under the new name German Biobank Network, which will also incorporate the German Biobank Alliance (GBA).
As part of the GBA and a member of the GBN, BioBank Dresden is also directly affected by this development. In addition, we are currently actively involved in the NUM study NAPKON NUKLEUS, which further underlines our close ties to the university medical research landscape.
Integration into the NUM will create new opportunities for collaboration - for example through even closer links with the data integration centers - and simplify access to quality-assured biosamples and associated data for researchers throughout Germany. This is an important step towards sustainable and future-oriented structures in biomedical research.
We are delighted to be pursuing this path together with our partners in the German Biobank Network and the NUM.
The complete press release can be found here.
----------------------------------------------------------------------------------------------------------------------------------
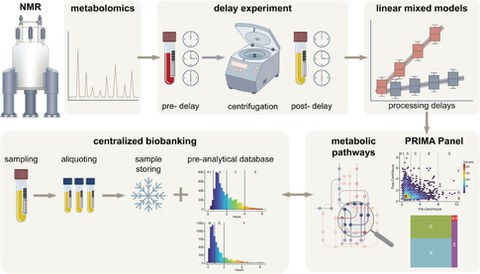
Exciting news from BioBank Dresden (BBD): Our first own publication has been published!
In the study entitled "Validating Centralized Biobanking Workflows for NMR Metabolomics Using the PRIMA Panel", our researchers validate centralized biobanking workflows for NMR metabolomics using the PRIMA panel. This work represents a significant advance in the standardization and quality assurance of biobanking processes and underlines BBD's commitment to research excellence. The full study can be found here.
----------------------------------------------------------------------------------------------------------------------------------
We are pleased to report on the successful 3rd meeting of our Scientific Advisory Board (SAB), which has been held regularly since 2019. This time, the focus was on the exchange with Dr. med. Michael Kiehntopf (3rd from left) , Director of the Institute for Clinical Chemistry and Laboratory Medicine in Jena and the integrated biobank. His concept has many parallels to our approach, which led to an inspiring and insightful dialog. Mr. Kiehntopf is a leading expert in biospecimen quality management, with numerous publications and active membership in international committees and biobank consortia such as ESBB and BBMRI.
In addition, Prof. Dr. Thomas Illig (5th from left), Head of the HUB Hannover and central stakeholder in networks such as NUM, and Prof. Dr. Roland Jahns (4th from left), Head of the IDBW Würzburg and Chairman of the Würzburg Ethics Committee, enriched the discussions with their extensive knowledge and experience.
The exchange was characterized by constructive suggestions for improvement and inspiring perspectives that further strengthen and advance our concept.
We would like to thank all participants for their commitment and look forward to the next steps!
----------------------------------------------------------------------------------------------------------------------------------
In the sixth video entitled "Accreditation in Focus - Quality Management in Biobanks", Dr. Heidi Altmann, PD Dr. Dr. Michael Kiehntopf and Prof. Dr. Thomas Illig discuss the GBN quality program, in particular the "Friendly Audits" and the EQA schemes. Michael Kiehntopf begins by emphasizing: "Our aim is to ensure that all samples in the network are of consistent and standardized quality." Heidi Altmann explains the role of the Friendly Audits, which help the biobanks of the German Biobank Alliance (GBA) with accreditation in accordance with DIN EN ISO 20387: "The auditors support our quality management in a very constructive way." Thomas Illig, Head of the recently accredited Hannover Unified Biobank (HUB), summarizes: "We couldn't have done it without GBN."
----------------------------------------------------------------------------------------------------------------------------------
The German Biobank Node (GBN), BBMRI.at and the Swiss Biobanking Platform (SBP), all three "National Nodes" of the European biobanking organization BBMRI-ERIC, have jointly published a new film. It highlights the important role of professional biobanks for research and the promotion of new discoveries in medicine.
With the slogan "Professional biobanks can make your life easier", the two-minute animated film is aimed at researchers and encourages them to work with professional biobanks. The biobanks in the GBN, BBMRI.at and SBP networks offer comprehensive advice and support, work according to international standards to ensure high sample quality, adhere to strict safety protocols and are committed to the long-term success of research.
The resulting film not only serves to provide information, but is also an expression of the intensive cooperation between the "National Nodes" - in line with the founding idea of BBMRI-ERIC. Likewise, as the film shows, collaboration between researchers and biobanks can accelerate research and pave the way to a healthier future.
----------------------------------------------------------------------------------------------------------------------------------
Change in the management of CCP-Bio: Dr. Heidi Altmann and Dr. Katja Steiger take over
In the second quarter of 2024, a significant change in leadership took place within the DKTK Clinical Communication Platform (CCP): Dr. Heidi Altmann and Dr. Katja Steiger have taken over the management of CCP-Bio, the area for networked biobanking, from Prof. Michael Hummel. This handover marks a new phase for the platform, which focuses on optimizing access to biosamples. You can find the complete article here.
----------------------------------------------------------------------------------------------------------------------------------
As part of its anniversary year 2024, the German Biobank Node (GBN) presents a special video series. This series offers insights into the GBN's past successes and activities. As one of the representatives, our spokesperson Dr. Heidi Altmann also has her say.
----------------------------------------------------------------------------------------------------------------------------------
New project launched:
In a delightful partnership with a pioneering environmentally conscious project, we are pleased to announce that LVL has now officially started production of its fully recycled rack series.
----------------------------------------------------------------------------------------------------------------------------------
We had two significant site visits in March:
The esteemed team around Dr. Nadine Volk from NCT Cell and Liquid Biobank // Heidelberg took the opportunity to evaluate our biobank infrastructure with regard to the automated workflow of PBMC isolation (isolation of cells from blood and bone marrow). The overall goal is to provide a homogeneous sample collection for national cancer research across different sites. To this end, the PBMC biobanking process (automated isolation, freezing and storage) was implemented in parallel to the process in Heidelberg. The visit enabled a fruitful exchange and intensive discussions about possible solutions to challenges that arise and potential opportunities for cooperation.
This was followed by a visit from the team led by Dr. Diana Debreceni, Head of the Biobank Facility at the Center for Molecular Fingerprinting at the University of Szeged in Hungary, and Dr. Frank Fleischmann from the Max Planck Institute of Quantum Optics in Garching. The focus was on a tour of our infrastructure, accompanied by lively discussions about the biobanking workflow with a particular focus on automated storage. The background to this is the goal of establishing a national biobanking infrastructure in Hungary for the epidemiological biobanking of healthy samples. The visit was characterized by a lively exchange on topics such as process planning, system configuration, commissioning, validations, troubleshooting and data management in the biobanking context.
At the 11th National Biobanking Symposium in May last year, LVL Technologies GmbH & Co. KG caused a stir by raffling off a custom-made bench with the striking "Biobank" lettering, made with LVL Tubes. This bench, handmade by a carpenter, quickly found a lucky winner in our Quality and Project Manager, Dr. Susann Witt.
After an exciting wait, the bench was placed in our nitrogen storage facility on 20.03.2024 with due ceremony. Mr. Tobias Häßner from LVL Technologies was personally present to bring the bench to its new location and symbolically hand it over to BioBank Dresden. This gesture underlines not only the importance of cooperation between industry and research institutions, but also the innovative strength and sense of community that characterizes the biobank community.
----------------------------------------------------------------------------------------------------------------------------------
Press Release
Ten years of the German Biobank Node (GBN) - A decade of progress in biomedical research
Berlin, 17.01.2024. Human biospecimens stored in biobanks and linked data help to detect diseases at an early stage and treat them successfully. The German Biobank Node (GBN), an umbrella organization for academic biobanks in Germany, was founded to make these valuable resources more easily accessible for national and international research while ensuring the highest quality standards. This year, the GBN is celebrating its tenth anniversary.
Dr. Heidi Altmann comments: "The future of quality-oriented biobanking lies in strong cooperation at national and European level. The work of the GBN helps to accelerate research and develop innovative therapies."
You can read the full press release here. A short article was also published in the current Ärztezeitung.
----------------------------------------------------------------------------------------------------------------------------------
A starter kit is now available online via GBA, which can be used as a tool for setting up biobanks. This was co-developed by our BBD spokesperson Dr. Heid Altmann.
----------------------------------------------------------------------------------------------------------------------------------
Employees of different biobank sites of the German Biobank Alliance (GBA ) have the opportunity to audit each other once a year in the form of so-called "friendly audits". These audits support quality management by ensuring that the organization's processes, requirements and guidelines meet the prescribed standards. On 18.12.2023, we successfully conducted and completed our first GBA friendly audit.
----------------------------------------------------------------------------------------------------------------------------------
Dr. Heidi Altmann was elected to the Steering Committee of the German Biobank Alliance for 2024-2025. More details here.
----------------------------------------------------------------------------------------------------------------------------------
Our quality manager Dr. Susann Witt has successfully completed her training and is now an official auditor for German Biobank Node.
----------------------------------------------------------------------------------------------------------------------------------
3Sat showed great insights into the work of our biobank in an exciting documentary on"Collecting, storing and preserving" on 16.12.2023.
The documentary is still available to watch in the media library.
----------------------------------------------------------------------------------------------------------------------------------
From August 21, 2023 to August 2024, you will find the exhibition "From Shadow to Light" in House 136 of the NCT Dresden.
In this context, Julia Schmelzer is showing, among other things, a film project that was created in collaboration with BioBank Dresden and the NCT/UCC and deals with modern cancer medicine and research.
The camera takes the viewer through parts of our DILB, the sober reality of examination equipment and laboratories, while at the same time delving deep into the microworld of microbiological and organic details.